|
The more orbitals there are, the further away the electrons are from the nucleus.The size of an atom’s radius is also affected by the number of orbitals surrounding the nucleus. This force increases as the number of protons in the nucleus increases.Īs a result, atoms with more protons tend to have smaller radii than those with fewer protons. The larger the nucleus, the greater the attractive force between the protons and the electrons in the orbitals. The radius of an atom is determined by the size of its nucleus. What factors affect the size of an atom’s radius of the largest element In general, the size of an atom is an important determinant of its physical and chemical properties. They also tend to have higher melting and boiling points. For example, large atoms tend to be denser than small atoms. The size of an atom also affects its properties. 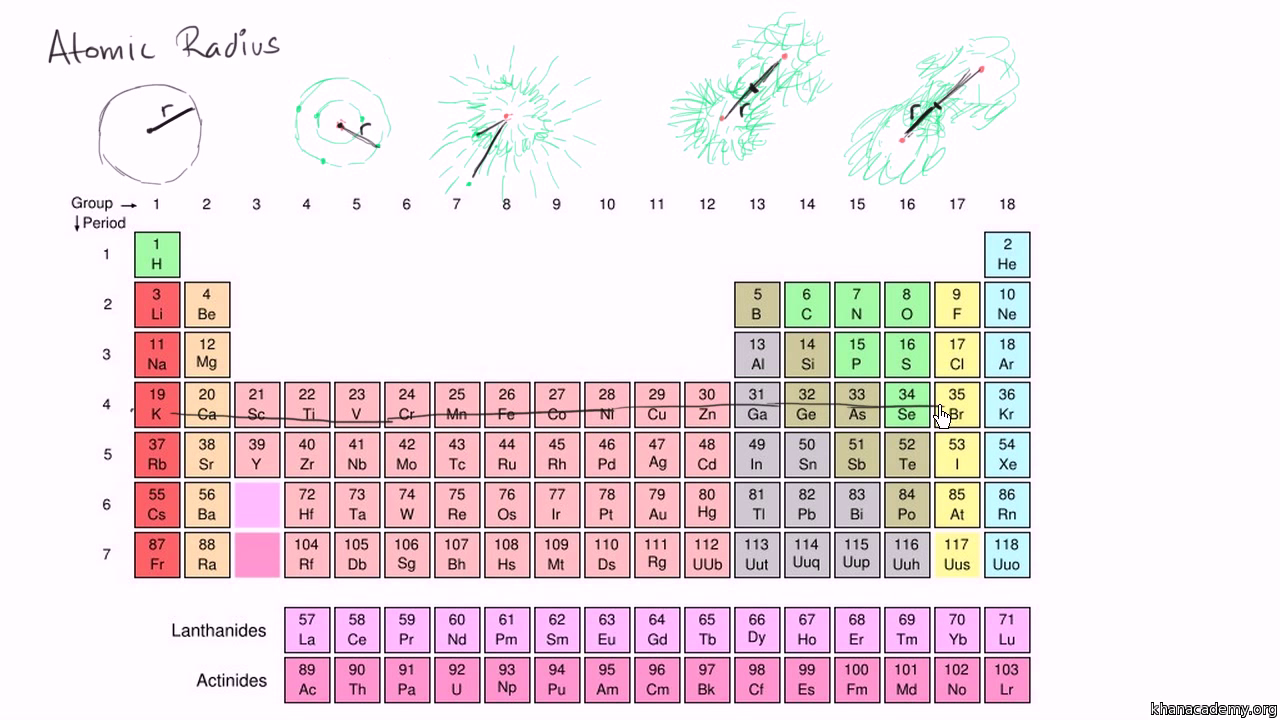
Atoms with more neutrons are typically larger than atoms with fewer neutrons.However, the size of an atom also depends on the presence of neutrons.The larger the number of protons, the larger the atom.The size of an atom is determined by the number of protons in its nucleus. Ultimately, the atomic radius of an element can be thought of as an average distance, and it is generally represented by either a single value or a range of values. However, this model is not always accurate, and other methods may be used in certain cases. Most often, atomic radius is calculated using the Bohr model of the atom, which treats electrons as orbiting the nucleus in a series of defined orbits. The difficult part of this calculation is that electrons are constantly moving, so there is no definitive answer. The atomic radius of an element is the distance from the center of the nucleus to the outermost electron in the atom. How do you calculate the atomic radius of the largest element? This makes francium about five times larger than hydrogen.įrancium is also one of the heaviest known elements its atoms weigh nearly twice as much as hydrogen atoms.ĭespite its large size, francium is rare and has only been found in trace amounts in the laboratory. In reality the electrons tend to screen each other somewhat from the inner positive charge and so the size stays about the same.Which element has the largest atomic radius?īy comparison, the diameter of a hydrogen atom is about 53 picometers. You might think that as you add more protons (and thus more positive charge), the electrons would be attracted more strongly to the inner nucleus and hence the atom would shrink. The tightly packed nucleus is 100,000 times smaller than the electron cloud. An atom is about 10 -8 centimeters in size (meaning that 100 million of them would fit side-by-side within one centimeter). Atoms are composed of a nucleus (where the positively charged protons and uncharged neutrons reside) surrounded by a cloud of orbiting negatively charged electrons. On the other hand, if you are speaking of size, then atoms are all about the same size whether it's a hydrogen atom (the simplest and least massive with one proton and one electron) or a lead atom (with 82 protons, 82 electrons and 125 neutrons). Very massive atoms such as nobelium and lawrencium have lifetimes of only a few seconds. However, for very massive atoms, the force holding them together becomes unstable and they tend to break apart (a phenomenon known as radioactive decay). As you add more protons and neutrons, the mass increases.  
Since electrons are about 2000 times less massive than protons (and neutrons), then the mass of an atom is mostly from the protons and neutrons. If by "biggest" and "smallest", you mean mass (which is a measure of how much matter is there), then the smallest is the hydrogen atom with one proton and one electron. What is the biggest atom? The smallest atom?
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
